What is Entresto (sacubitril/valsartan) for?
Entresto (sacubitril/valsartan) is a neprilysin inhibitor and angiotensin II receptor blocker combination used to treat:
- adults with long-lasting (chronic) heart failure to help reduce the risk of death and hospitalization.
- pediatric patients aged one year and older with symptomatic heart failure who have systemic left ventricular systolic dysfunction
The medicine works better in patients whose heart cannot pump a normal amount of blood to the body.
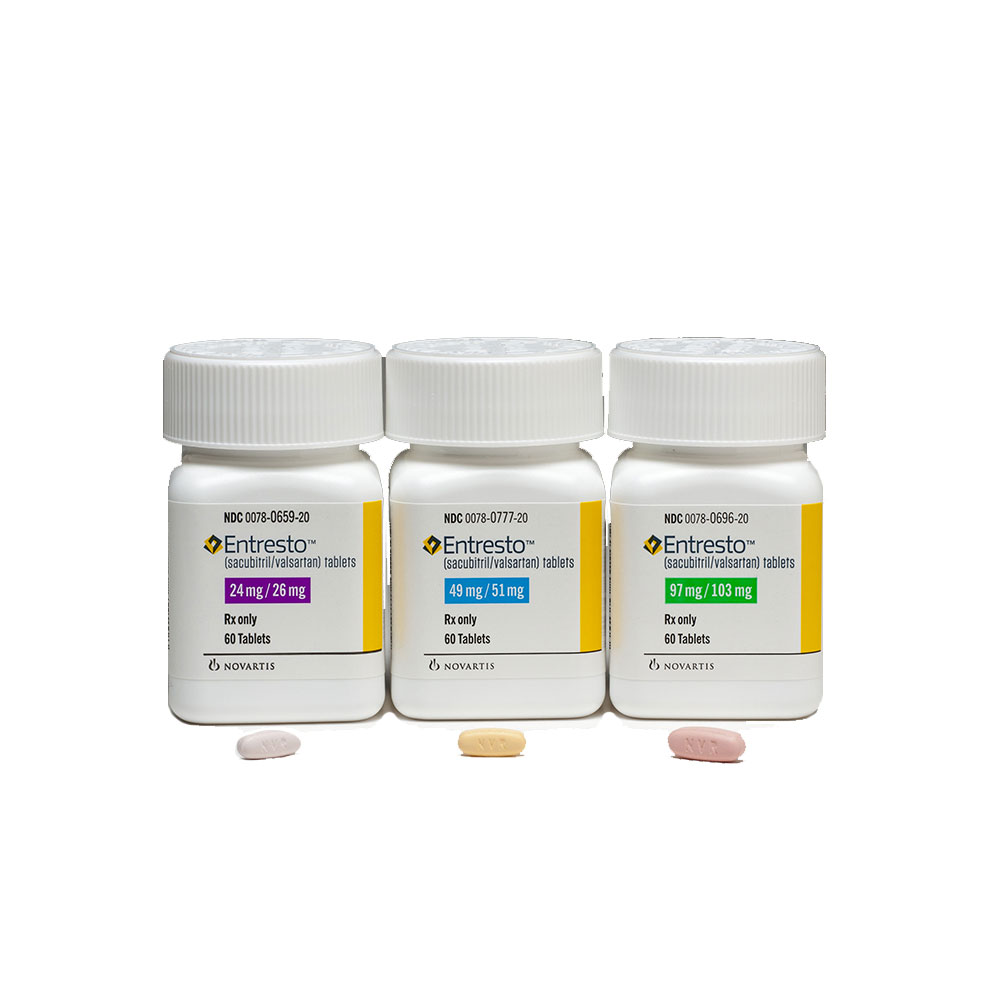
It is available in tablet form, each containing 24 mg/26 mg, 49 mg/51 mg, or 97 mg/103 mg sacubitril/valsartan.
How does Entresto (sacubitril/valsartan) work?
The two active ingredients in Entresto, sacubitril and valsartan, have different effects on the heart.
Sacubitril prevents the breakdown of natriuretic peptides that are made in the body. These peptides make sure that natrium and water are excreted via the urine, which unloads the burden on the heart. Natriuretic peptides also lower blood pressure and protect the heart against scarring (a process called fibrosis) that happens during heart failure.
Valsartan is an angiotensin II receptor blocker. It blocks the hormone angiotensin II, which may have damaging effects in patients with heart failure. By blocking its receptor, valsartan can reduce the damaging effects of angiotensin II on the heart. It also reduces blood pressure by making sure that blood vessels can dilate.
Where has Entresto (sacubitril/valsartan) been approved?
Entresto (sacubitril/valsartan) was approved by:
- The Food and Drug Administration (FDA), USA:
- On July 7, 2015 to treat heart failure in adults.
- On October 1, 2019 to treat pediatric heart failure.
- On February 16, 2021 to treat chronic heart failure.
- The European Medicines Agency (EMA):
- On November 19, 2015 to treat heart failure in adults.
- On September 11, 2019 to treat pediatric heart failure.
- National Medical Products Administration (NMPA), China:
- In 2017 to treat heart failure.
- On June 10, 2021 to treat hypertension.
Entresto is approved in 115 countries worldwide for the treatment of heart failure with reduced ejection fraction.
Please note that this medicine may have also been approved in other regions than the ones we’ve listed. If you have a question about its approval in a specific country feel free to contact our support team.
How is Entresto (sacubitril/valsartan) taken?
The standard dosage is:
- The recommended starting dose is one tablet of Entresto 49 mg/51 mg twice daily
- After 2 to 4 weeks, the doses are then doubled to 97 mg / 103 mg twice daily.
The doctor may choose lower doses for certain patients.
It is usually used in combination with other heart failure therapies, in place of an ACE inhibitor or other ARB. It is a first-choice treatment for patients with heart failure with reduced ejection fraction.
Entresto (sacubitril/valsartan) may affect the way other medicines work, and other medicines may affect how this medicine works, so it is important that you inform your doctor about all medicines you take.
Warning: you should not take an ACE inhibitor medicine and Entresto together. Do not take this medicine when you have a history of angioedema or liver disease.
Kidney function and blood potassium levels will be monitored in patients.
Complete information about Entresto (sacubitril/valsartan) dosage (modifications) and administration can be found in the official prescribing information listed in our references section.
Note: Please consult with your treating doctor for personalised dosing.
Are there any known adverse reactions or side effects of Entresto (sacubitril/valsartan)?
Common adverse reactions
The most common side effects (≥5% of patients) listed in the prescribing information include:
- Low blood pressure (hypotension)
- Kidney problems
- Increased levels of potassium in your blood (hyperkalemia)
- Dizziness
Serious adverse reactions
The serious adverse reactions listed in the prescribing information include:
- Serious allergic reactions
Use in a specific population
Entresto (sacubitril/valsartan) can be fatal for a fetus, it is advised to avoid pregnancies and breastfeeding.
For a comprehensive list of side effects and adverse reactions please refer to the official prescribing information.
References
1. Full prescribing information [FDA]: Entresto (sacubitril/valsartan) [PDF]
Novartis, Jul 2015
2. How Entresto works
Novartis, cited Aug 2021
3. Entresto product information
EMA, Nov 2015


Reviews
There are no reviews yet.